Mitochondrial Disease Therapeutic Pipeline
Several treatments for mitochondrial diseases are already making their way along this path. Now, mito patients and their families will be able to see the treatment approaches being developed, where they are in the development pipeline, and what mitochondrial diseases they target.
Conditions in the Pipeline
- Barth Syndrome
- KSS-CPEO
- Leigh Syndrome
- LHON
- MELAS
- MERRF
- MIDD
- Primary Mitochondrial Myopathies (PMM)
- Pearson Syndrome
- POLG
- Pyruvate Dehydrogenase Complex Deficiency (PDCD)
- TK2 Deficiency
Pre-Clinical Development
Phase I
Phase II
Phase III
Regulatory Approval
Phase II/III tests how well a new treatment works and compares the new treatment with a standard treatment.
Barth Syndrome
Elamipretide (SS-31/MTP-131)
Stealth BioTherapeutics
Name:
FORZINITY
Status:
Approved by FDA

CoQ10 deficiency
BPM31510IV
BPG Bio, Inc.
Status:
Study in Development

KSS-CPEO
KL1333
Abliva/Pharming
Study Name:
FALCON
Status:
Actively Recruiting

Leigh Syndrome
MNV-201
Minovia
Status:
Progressing to clinical development

NV354
Abliva/Pharming
Status:
Progressing to clinical development

LHON
Idebenone
Chiesi Global Rare Diseases
Status:
Issued Complete Response Letter in March 2026; Conversations with FDA continue

LUMEVOQ (GS010)
GenSight Biologics
Study Name:
REFLECT
Status:
Study active but not recruiting

NR082
Neurophth Biotechnology
Status:
Actively Recruiting
(Site in China)
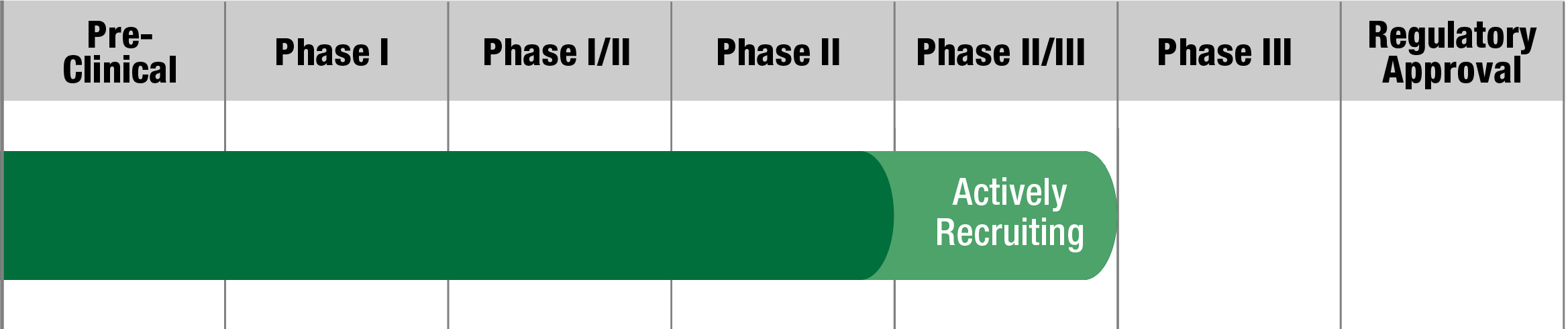
MELAS
OMT-28
Omeicos Therapeutics
Study Name:
PMD-OPTION
Status:
Fully Recruited
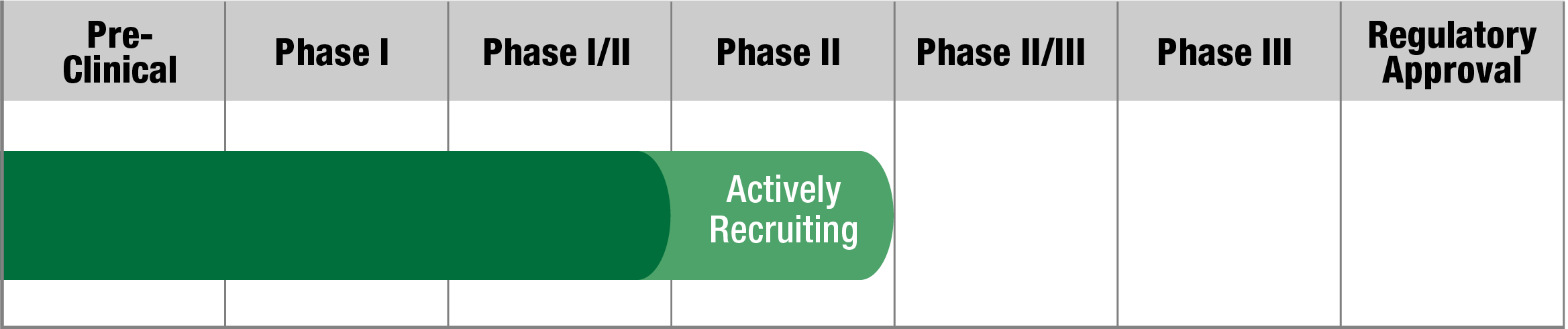
Zagociguat
Tisento Therapeutics
Study Name:
PRIZM
Status:
Actively Recruiting
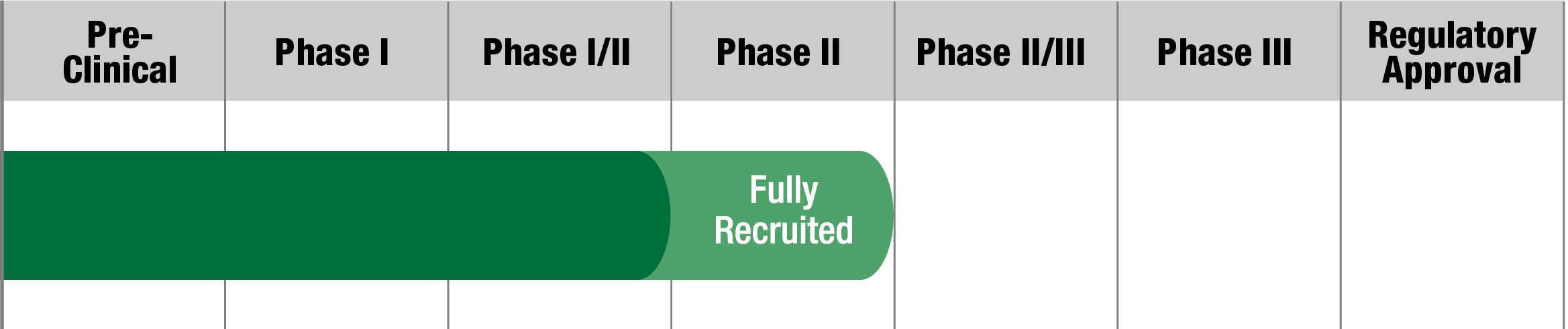
Sonlicromanol (KH176)
Khondrion
Study Name:
KHENERFIN
Status:
Active, Not Yet Recruiting

KL1333
Abliva/Pharming
Study Name:
FALCON
Status:
Actively Recruiting

MERRF
OMT-28
Omeicos Therapeutics
Study Name:
PMD-OPTION
Status:
Fully Recruited

KL1333
Abliva/Pharming
Study Name:
FALCON
Status:
Actively Recruiting

MIDD
OMT-28
Omeicos Therapeutics
Study Name:
PMD-OPTION
Status:
Fully Recruited

Sonlicromanol (KH176)
Khondrion
Study Name:
KHENERFIN
Status:
Active, Not Yet Recruiting

KL1333
Abliva/Pharming
Study Name:
FALCON
Status:
Actively Recruiting

Pearson Syndrome
MNV-201
Minovia
Status:
Actively Recruiting
(Site in Israel)

PDCD
SL-1009
(Sodium dichloroacetate)
Saol
Status:
Under Regulatory Review

Primary Mitochondrial Myopathies (PMM)
Elamipretide (SS-31/MTP-131)
Stealth BioTherapeutics
Study Name:
NuPower
Status:
Fully Recruited

OMT-28
Omeicos Therapeutics
Study Name:
PMD-OPTION
Status:
Fully Recruited

POLG
PX578
Pretzel Therapeutics
Status:
Phase 1 – Safety Evaluation

TK2 Deficiency
MT1621
UCB
Name:
Kygevvi
Status:
Approved by FDA 11/3/25

